 The valence electrons are held closer towards the nucleus of the atom. This means that the nucleus attracts the electrons more strongly, pulling the atom's shell closer to the nucleus. 
The effect of increasing proton number is greater than that of the increasing electron number therefore, there is a greater nuclear attraction. However, at the same time, protons are being added to the nucleus, making it more positively charged. This is because, within a period or family of elements, all electrons are added to the same shell. Atomic radius patterns are observed throughout the periodic table.Ītomic size gradually decreases from left to right across a period of elements. The covalent radii of these molecules are often referred to as atomic radii. Nevertheless, it is possible for a vast majority of elements to form covalent molecules in which two like atoms are held together by a single covalent bond. Some are bound by covalent bonds in molecules, some are attracted to each other in ionic crystals, and others are held in metallic crystals. However, this idea is complicated by the fact that not all atoms are normally bound together in the same way. The atomic radius is one-half the distance between the nuclei of two atoms (just like a radius is half the diameter of a circle). This is caused by the increase in atomic radius. Electron affinity decreases from top to bottom within a group.This is caused by the decrease in atomic radius. Electron affinity increases from left to right within a period.This causes the electron to move closer to the nucleus, thus increasing the electron affinity from left to right across a period. Moving from left to right across a period, atoms become smaller as the forces of attraction become stronger. IUPAC involvement covers various aspects of the table and data that it unveils, and several reports and recommendations, some quite recent, attest of that input. 
With a larger distance between the negatively-charged electron and the positively-charged nucleus, the force of attraction is relatively weaker. By virtue of its work in relation with the chemical elements, IUPAC can dispense a periodic table that is up-to-date. This means that an added electron is further away from the atom's nucleus compared with its position in the smaller atom. In rechargeable batteries, such as many lithium-ion batteries, this chemical process is reversible and the internal structure different which allows the batteries to be recharged.ĭue to the ionic properties of salt water, scientists are now striving to exploit the ionic electricity-generating potential of salinity gradients where salt water and fresh water mix as a green source of energy generation for the future.\( \newcommand\): Periodic Table showing Electron Affinity TrendĮlectron affinity generally decreases down a group of elements because each atom is larger than the atom above it (this is the atomic radius trend, discussed below). The zinc anode also acts as the battery’s container in zinc-carbon batteries so as it oxidizes during use, the contents can start to leak over time.įigure 4: A zinc-carbon dry cell battery (left) and alkaline battery (right). In single use, dry cell batteries, zinc is commonly used as the anode whilst manganese dioxide is a popular choice for the electrolyte cathode. As this ionic substance reacts with the electrodes it generates electrical current. In between the electrodes is an electrolyte liquid or gel that contains charged particles – ions. 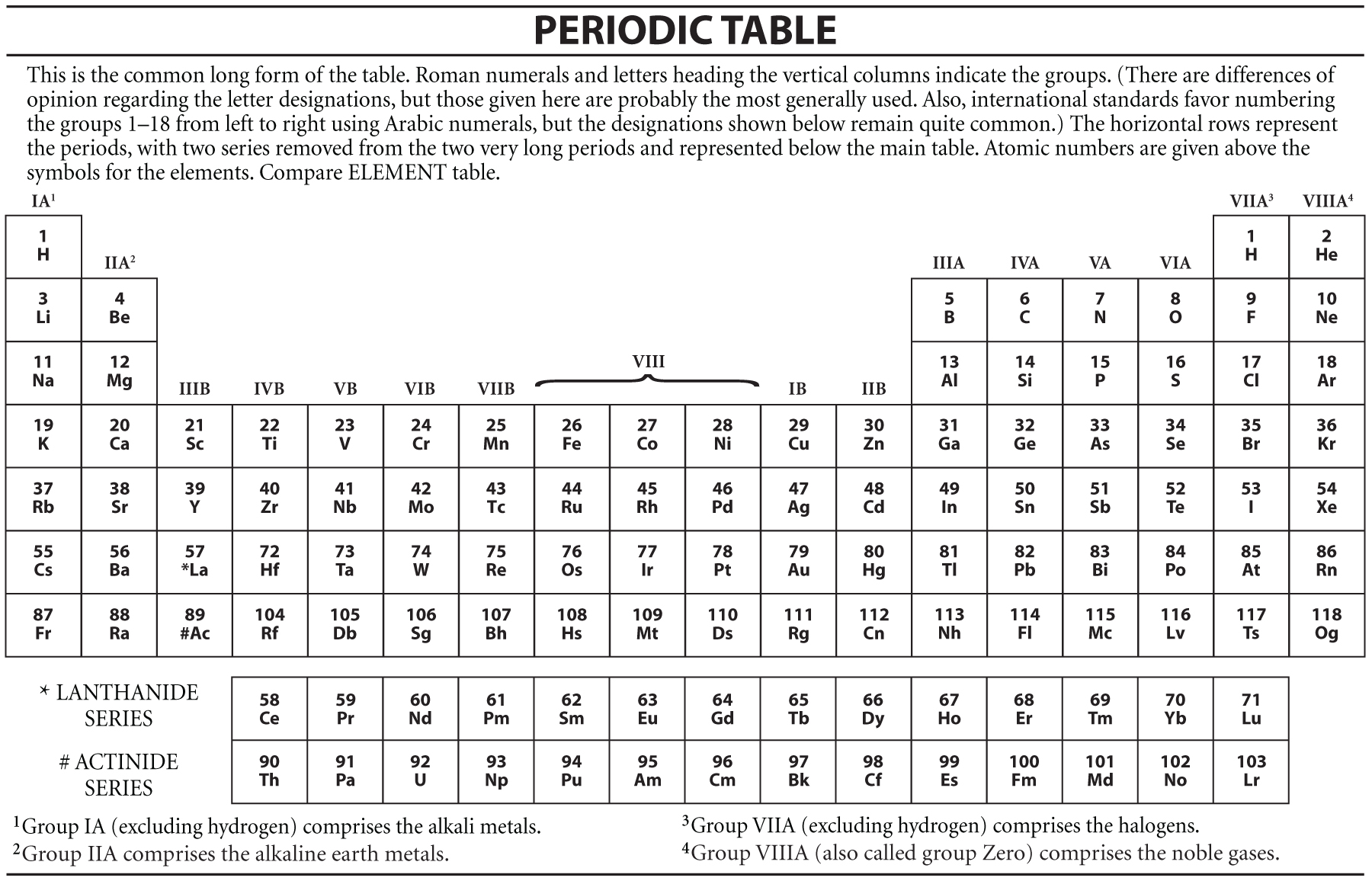
Batteries have two electrodes made of conductive material, the cathode which is the positive end where the electrical current leaves/electrons enter, and the anode where the electrical current enters/ electrons leave. Ionic properties are central to the function of batteries too. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
